 |
Ben started his PhD in Paunov Research Group in October 2013 funded by EPSRC industrial CASE award. He graduated with PhD in 2017. After a post-doc at the University of Maryland, he is now doing a second post-doc at the University of Delaware.PhD supervisors: Prof. Vesselin Paunov (PI) and Dr Tommy Horozov (Co-PI) |
Research project 1:
Novel Methods for Preparation of Hierarchically Porous Materials
Materials with engineered porosity have applications in structuring of food products, pharmaceuticals and household formulations. The project involve a variety of templating techniques and porogenic agents to produce hierarchically porous materials with more than one level of porosity. We developed novel hierarchically porous materials prepared by using two recently developed approaches based on hydrogel templating technique using hydrogel solutions and slurries of hydrogel beads. Materials with hierarchical porosity were produced by using a dual templating technique with two different sized hydrogel beads. As a proof of principle we applied these methods to two different types of matrix materials: cement and gypsum.
We also developed a hybrid method for fabrication of hierarchically porous materials that combines the hydrogel templating technique with the viscous trapping technique, allowing us to produce materials with dual porosity. The methods used were able to achieve composite porosities between 50 % to 80 % with minimal volume contraction upon drying. We also study the mechanical properties and sound adsorbing properties of these porous materials. The results showed that decreased pore size from smaller hydrogel beads gave increased the porous composite compressional strength and elastic modulus. For porous materials with pores induced by different hydrogel solutions, there was noticeable differences in compressional strength. We found that for hierarchically porous materials, the compressional strength changed when varying the ratios of micropores to mesopores. The sound adsorption coefficient of the hierarchically porous materials changed with changing the materials porosity but was not sensitive to the material pore size distribution.

Figure 1: Schematics of preparation of porous cement composite from cement slurry and hydrogel slurry. The pore size can be controlled by the average size of the hydrogel beads which is controlled by the blending conditions.

Figure 2: SEM image of a porous cement composite with dual porosity.
References
- Rutkevicius, M., Munusami, S.K., Watson, Z., Field, A.D., Salt, M., Stoyanov, S.D., Petkov, J., Mehl, G.H., Paunov, V.N., “Fabrication of novel lightweight composites by a hydrogel templating technique”, Res. Bull., 47 (2012) 980–986.
- Rutkevicius, M., Mehl, G.H., Qin, Q., Rubini, P.A., Stoyanov, S.D., Paunov, V.N., Sound absorption properties of porous composites fabricated by a hydrogel templating technique, Mater. Research, 28 (2013) 2409-2414.
- Rutkevicius, M., Austin, Z., Chalk, B., Mehl, G.H., Qin, Q., Rubini, P.A., Stoyanov, S.D., Paunov, V.N., Sound absorption of porous cement composites: effects of the porosity and the pore size, Materials Science, 50 (2015) 3495-3503.
- Thompson, B.R. Horozov, T.S., Stoyanov, S.D., Paunov, V.N., Hierarchically porous composites fabricated by hydrogel templating and viscous trapping techniques, Materials & Design, 137 (2018) 384-393.
- Thompson, B.R., Taylor, B.L., Qin, Q., Stoyanov, S.D., Horozov, T.S., Paunov, V.N., Sound transmission loss of hierarchically porous composites produced by hydrogel templating and viscous trapping techniques, Materials Chemistry Frontiers, 1 (2017) 2627-2637.
Research project 2:
Ultra Melt-Resistant Hydrogels from Food Grade Carbohydrates
We developed a binary hydrogel system made from two food grade biopolymers, agar and methylcellulose (Agar-MC) which does not require addition of salt for gelation to occur and has very unusual rheological and thermal properties (see Ref.1). We found that the storage modulus of the Agar-MC hydrogel far exceeds those of hydrogels from the individual components. In addition, the Agar-MC hydrogel has enhanced mechanical properties over the temperature range 25-85 °C and a maximum storage modulus at 55 °C when the concentration of methylcellulose was 0.75% w/v or higher (see Fig.2). This is explained by a sol-gel phase transition of the methylcellulose upon heating as supported by differential scanning calorimetry (DSC) measurements. Above the melting point of agar, the storage modulus of Agar-MC hydrogel decreases but is still an elastic hydrogel with mechanical properties dominated by the MC gelation. By varying the mixing ratio of the two polymers, agar and MC, it was possible to engineer a food grade hydrogel of controlled mechanical properties and thermal response. SEM imaging of flash-frozen and freeze-dried samples revealed that the Agar-MC hydrogel contains two different types of heterogeneous regions of distinct microstructures (Fig. 1). The latter was also tested for its stability towards heat treatment which showed that upon heating to temperatures above 120 °C its structure was retained without melting. The produced highly thermally stable hydrogel shows melt resistance which may find application in high temperature food processing and materials templating.
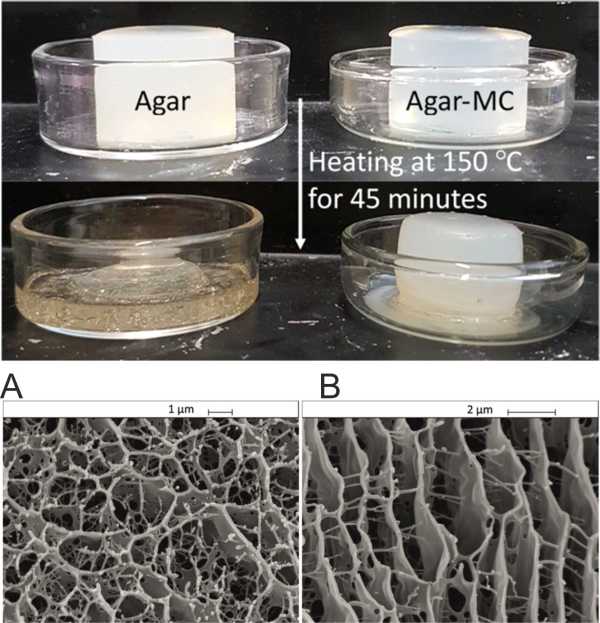
Fig. 1. TOP: Samples of agar hydrogel (2.0% w/v) and the agar-MC hydrogel (1.0% w/v – 1.0% w/v) before (top) and after (bottom) heating in a furnace at 150 °C for 45 minutes. BOTTOM: Freeeze dries samples of (A) Agar hydrogel, (B) Agar-Methyl cellulose hydrogel.

Fig. 2. The storage modulus of 2.0% w/v agar hydrogel, 2.0% w/v MC hydrogel and 1% w/v : 1% w/v Agar:MC hydrogel as a function of the temperature. G’ is measured at 100 Pa.
References
- Thompson, B.R. Horozov, T.S., Stoyanov, S.D., Paunov, V.N., An ultra melt-resistant hydrogel from food grade carbohydrates, RSC Advances, 7 (2017) 45535-45544.
Project 3:
Structuring and calorie control of bakery products by templating with melt-resistant hydrogel beads
We report the use of a temperature insensitive, food-grade hydrogel to reduce the caloric density of pancakes that were prepared at temperatures much higher than the boiling point of water. This cheap, facile method utilises a mixed agar-methylcellulose hydrogel, which was blended to produce slurry of hydrogel microbeads (see Fig.1). The pancake batter was mixed with a controlled volume percentage of slurry of hydrogel beads and cooked. From calorimetry experiments, the composites were found to have a reduced caloric density that reflects the volume percentage of hydrogel beads mixed with the batter. Using this procedure, we were able to reduce the caloric density of pancakes by up to 23 ± 3% when the volume percentage of hydrogel beads initially used was 25%.

Fig. 1. TOP: Schematics of the templating of batter with hydrogel slurries of highly melt-resistant hydrogel. BOTTOM: SEM images of freeze-dried pancake (A) and pancake-hydrogel composites (B). The composites were produced with an initial volume percentage of 17.5% of Agar-MC hydrogel beads (1% w/v – 1% w/v). Scale bars are 100 µm.
The method is not limited to pancakes and could potentially be applied to various other food products. The structure and morphology of the freeze-dried pancakes and pancake-hydrogel composites were investigated and pores of a similar size to the hydrogel beads were found, confirming the gel beads maintained their structure during the cooking process (Fig.2). There is scope for further development of this method by the encapsulation of nutritionally beneficial or flavor enhancing ingredients within the hydrogel beads.
Fig. 2. LEFT: An optical photograph of a pancake (A) and pancake-hydrogel composites initially containing (B) 10%, (C) 17.5% and (D) 25% by volume slurry of A-MC hydrogel beads. They were prepared by mixing a controlled volume percentage of slurry of hydrogel beads with pancake batter, then heating at 170 °C for 4 minutes on each side. RIGHT: Further mass reduction of the pancake-hydrogel composites compared to the pancake control sample due to the presence of hydrogel beads.
References
- Thompson, B.R. Horozov, T.S., Stoyanov, S.D., Paunov, V.N., Structuring and calorie control of bakery products by templating batter with ultra melt-resistant food-grade hydrogel beads, Food & Function, 8 (2017) 2967 – 2973.


