|
|
Ahmed Halbus started his PhD project in the Paunov Research Group at the Department of Chemistry of the University of Hull in October 2015. Ahmed graduated with PhD in Chemistry in 2019. Currently working as a lecturer at Babylon University, Iraq.PhD supervisors: Prof. Vesselin N. Paunov (PI) and Dr Tommy Horozov (Co-PI) |
Research project 1:
Self-grafting copper oxide nanoparticles with ultra-enhanced antibacterial action
Copper oxide nanoparticles (CuONPs) have been widely recognized as good antimicrobial agents but are heavily regulated due to environmental concerns of their post use. In this work, we have developed and tested a novel type of formulation for copper oxide (CuONPs) which have been functionalized with (3-Glycidyloxypropyl)trimethoxysilane (GLYMO) to allow further covalent coupling of 4-hydroxyphenylboronic acid (4-HPBA).
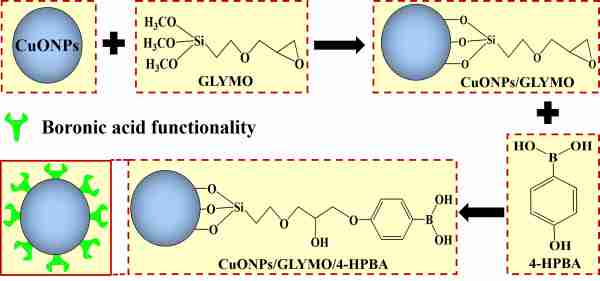
Figure 1. Schematics for preparation of self-grafting CuONPs.
As the boronic acid (BA) groups on the surface of CuONPs/GLYMO/4-HPBA can form reversible covalent bonds with the diol groups of glycoproteins on the bacterial cell surface, they can strongly bind to the cells walls resulting in a very strong enhancement of their antibacterial action which is not based on electrostatic adhesion [1].
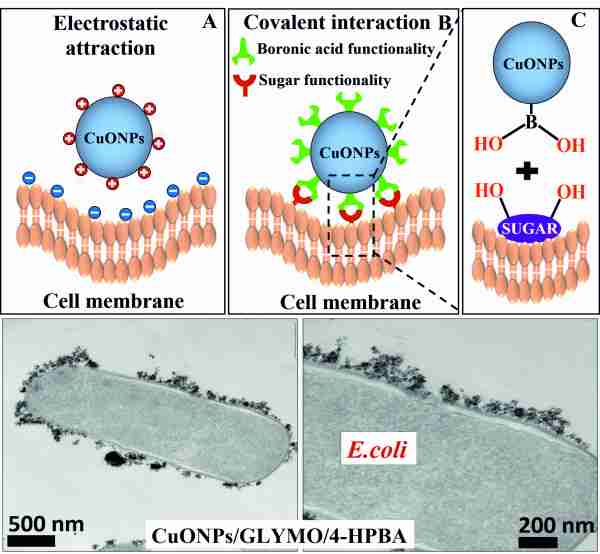
Figure 2. The interaction between the CuONPs with boronic acid surface functionality and the sugar groups on the surface of the bacterial cell wall leads to their attachment to the bacterial cell wall which strongly amplifies their antibacterial action. TEM images of E.coli after exposure to CuONPs/GLYMO/HPBA.
SEM and TEM imaging revealed that 4-HPBA-functionalized nanoparticles could accumulate more on the cell surface than non-functionalized ones (see Fig. 2). We demonstrate that the CuONPs with boronic acid surface functionality are far superior antibacterial agents compared to bare CuONPs. Our results showed that, the antibacterial impact of the 4-HPBA functionalized CuONPs on E.coli and R. rhodochrous is one order of magnitude higher than that of bare CuONPs or CuONPs/GLYMO (see Fig. 3).
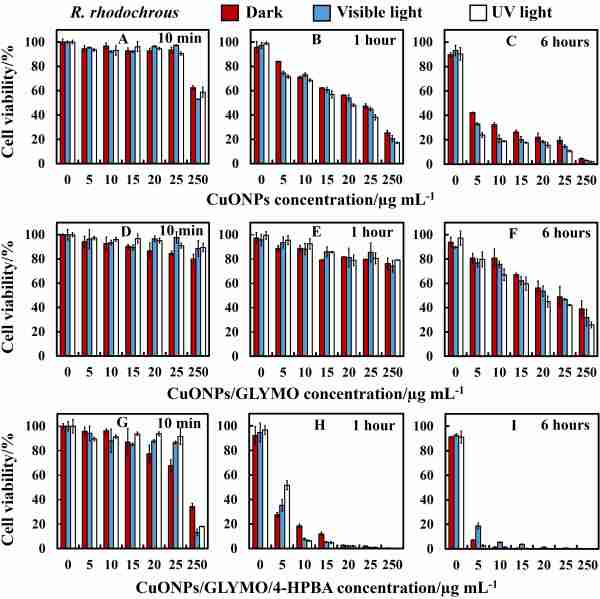
Figure 3. Comparison of the cell viobility of R. rhodochrous after exposure to bare CuONPs, CuONPs/GLYMO and CuONPs/GLYMO/HPBA.
We also observed a marked increase of the 4-HPBA functionalized CuONPs antibacterial action on these microorganisms at shorter incubation times compared with the bare CuONPs at the same conditions. We show that the cytotoxicity of CuONPs functionalized with 4-HPBA as an outer layer can be controlled by the concentration of glucose in the media and that the effect is reversible as glucose competes with the sugar residues on the bacterial cell walls for the BA-groups on the CuONPs. Our experiments with human keratinocytes exposure to CuONPs/GLYMO/4-HPBA indicated lack of measurable cytotoxicity at particle concentration which are effective as antibacterial agent for both bacteria. We envisage that formulations of CuONPs/GLYMO/4-HPBA can be used to drastically reduce the overall CuO concentration in antimicrobial formulations while strongly increasing their efficiency. We recently demonstrated that the same strategy works for anti-algal and anti-yeast formulations. [2]
References
1. Halbus, A.F., Horozov, T.S., Paunov, V.N., Strongly enhanced antibacterial action of copper oxide nanoparticles with boronic acid surface functionality, ACS Appl. Mater. Inter., 11 (2019) 12232–12243.
2. Halbus, A.F., Horozov, T.S., Paunov, V.N., Self-grafting copper oxide nanoparticles show a strong enhancement of their anti-algal and anti-yeast action, Nanoscale Advances, 1 (2019) 2323 – 2336.
Research project 2:
‘Ghost’ silica nanoparticles of ‘host’-inherited antimicrobial action
We fabricated surface-rough mesoporous silica nanoparticles (“ghost” SiO2NPs) by using composite mesoporous copper oxide nanoparticles (“host” CuONPs) as templates, which allowed us to mimic their surface morphology. The “host” CuONPs used here as templates, however, had a very high antibacterial effect, with or without functionalization. To evaluate the surface roughness effect on the “ghost” SiO2NPs antibacterial action, we functionalized them with (3-glycidyloxypropyl)trimethoxysilane (GLYMO) to permit additional covalent coupling of 4-hydroxyphenylboronic acid (4-HPBA). The diol groups on the bacterial membrane can form reversible covalent bonds with boronic acid (BA) groups on the “ghost” SiO2NPs surface and bind to the bacteria, resulting in a very strong amplification of their antibacterial activity.
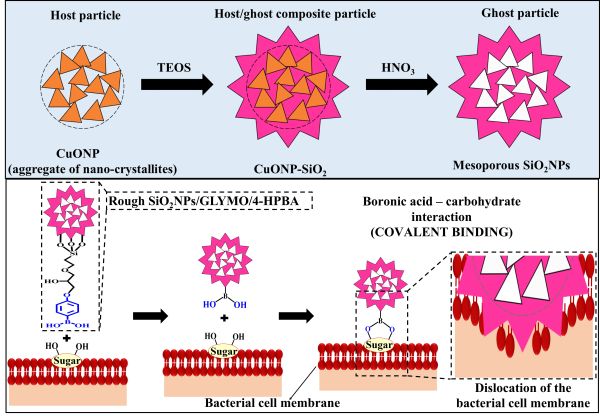
Figure 1. Top: schematic of the synthesis method of CuONPs/SiO2 (host/ghost composite particles) and surface-rough SiO2NPs (ghost particles) from CuONPs (host particles). Bottom: Schematics displaying the mechanism of self-grafting/covalent binding of the sugar groups expressed on the bacterial cell wall and boronic acid-functionalized surface-rough SiO2NPs (“ghost”).

Figure 2. TEM images of (A,E) bare CuONPs, which are aggregates of nanocrystallites (host), (B,F) SiO2-coated CuONPs producing a surface-rough SiO2NPs (host/ghost composite particles), (C,G) mesoporous surface-rough “ghost” SiO2NPs, and (D,H) smooth SiO2NPs of very similar particle size.
The BA-functionalized “ghost” SiO2NPs showed a very significant antibacterial effect as compared to smooth SiO2NPs of the same surface coating and particle size. We attribute this to the “ghost” SiO2NPs mesoporous surface morphology, which mimics to a certain extent those of the original mesoporous CuONPs used as templates for their preparation. We envisage that the “ghost” SiO2NPs effectively acquire some of the antibacterial properties from the “host” CuONPs, with the same functionality, despite being completely free of copper. The antibacterial effect of the functionalized “ghost” SiO2NPs/GLYMO/4-HPBA on Rhodococcus rhodochrous (R. rhodochrous) and Escherichia coli (E. coli) is much higher than that of the non-functionalized “ghost” SiO2NPs or the “ghost” SiO2NPs/GLYMO. We show that the rough surface morphology of the ‘ghost’ SiO2NPs, which upon covalent bonding between the rough SiO2NPs/GLYMO/HPBA and the bacterial cell membrane causes its impaling on the surface-rough features and produces membrane dislocation that kills the bacteria. This killing mechanism seems to work for both Gram-positive and Gram-negative bacteria.
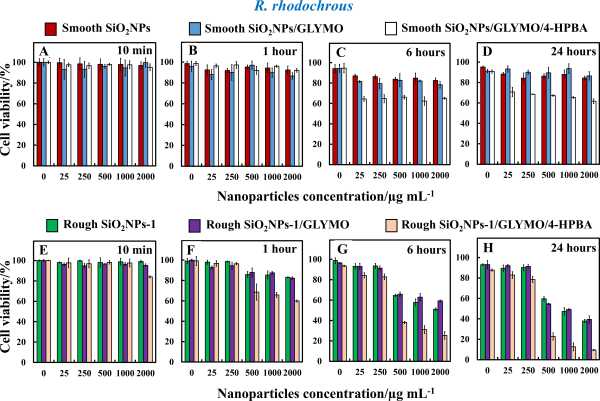
Figure 3: Cell viability of R. rhodochrous as a function of nanoparticle concentration with (A–D) smooth SiO2NPs, SiO2NPs/GLYMO, and SiO2NPs/GLYMO/4-HPBA. (E–H) “Ghost” SiO2NPs, “ghost” SiO2NPs/GLYMO, and “ghost” SiO2NPs/GLYMO/4-HPBA of various particle concentrations.
The results indicate that the combination of rough surface morphology and strong adhesion of the particle surface to the bacteria can make even benign material such as silica act as a strong antimicrobial agent. Additionally, our BA-functionalized nanoparticles (“ghost” SiO2NPs/GLYMO/4-HPBA) showed no detectable cytotoxic impact against human keratinocytes at particle concentrations, which are effective against bacteria.
References
1. Halbus, A.F., Horozov, T.S., Paunov, V.N., “Ghost” Silica Nanoparticles of “Host”-Inherited Antibacterial Action, ACS Appl. Mater. Interf., 11 (2019) 38519−38530.

