 |
Paul Weldrick started his PhD project in Paunov Research Group at the Department of Chemistry and Biochemistry of the University of Hull in October 2017. He graduated with PhD in Chemistry in April 2021. Currently he is doing a Post-doc at the University of Leeds.PhD supervisors: Prof. Vesselin N. Paunov (PI) and Prof Matthew Hardman (Co-PI) |
Research project 1:
Overcoming antibacterial resistance by antibiotic-loaded nanogel carriers with cationic surface functionality
Multidrug-resistant pathogens are prevalent in chronic wounds. There is an urgent need to develop novel antimicrobials and formulation strategies that can overcome antibiotic resistance and provide a safe alternative to traditional antibiotics. This work aimed to develop a novel nanocarrier for two cationic antibiotics, tetracycline hydrochloride and lincomycin hydrochloride which can potentially overcome antibiotic resistance. In this study, we report the use of surface functionalised polyacrylic copolymer nanogels as carriers for cationic antibiotics. These nanogels can encapsulate small cationic antimicrobial molecules and act as a drug delivery system. They were further functionalised with a biocompatible cationic polyelectrolyte, bPEI, to increase their affinity towards the negatively charged bacterial cell walls. These bPEI-coated nanocarrier-encapsulated antibiotics were assessed against a range of wound isolated pathogens, which had been shown through antimicrobial susceptibility testing (AST) to be resistant to tetracycline and lincomycin [1].
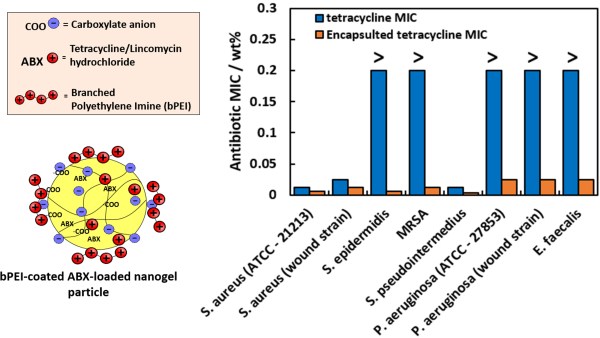
Fig. 1. LHS: Schematics of our nanogel particles loaded with a cationic antibiotic. RHS: Minimal Inhibitory Concentration (MIC) of free tetracyline and the nanogel-formulated tetracycline for a set of 8 different resistant and susceptible bacterial strains.
Our data reveal that bPEI-coated nanogels with encapsulated tetracycline or lincomycin displayed increased antimicrobial performance against selected wound-derived bacteria, including strains highly resistant to the free antibiotic in solution. Additionally, our nanocarrier-based antibiotics showed no detectable cytotoxic effect against human keratinocytes. We attribute the increase in the antimicrobial activity of the cationically functionalised antibiotic-loaded nanogel carriers to specific electrostatic adhesion to the microbial cell wall delivering a higher local antibiotic concentration, confirmed by scanning electron microscopy. Such a nanotechnology based approach may enhance the effectiveness of a wide variety of existing antibiotics, offering a potentially new mechanism to overcome antibiotic resistance.
References
Weldrick, P.J., Iveson, S., Hardman, M.J., Paunov, V.N., Breathing new life into old antibiotics: Overcoming antibacterial resistance by antibiotic-loaded nanogel carriers with cationic surface functionality, Nanoscale, 2019, 11, 10472-10485.
Research project 2:
Enhanced Clearing of Wound-Related Pathogenic Bacterial Biofilms
Biofilms are prevalent in chronic wounds and once formed are very hard to remove, which is associated with poor outcomes and high mortality rates. Biofilms are comprised of surface-attached bacteria embedded in an extracellular polymeric substance (EPS) matrix, which confers increased antibiotic resistance and host immune evasion. Therefore, disruption of this matrix is essential to tackle the biofilm-embedded bacteria. Here, we propose a novel nanotechnology to do this, based on protease-functionalized nanogel carriers of antibiotics [1]. Such active antibiotic nanocarriers, surface coated with the protease Alcalase 2.4 L FG, “digest” their way through the biofilm EPS matrix, reach the buried bacteria, and deliver a high dose of antibiotic directly on their cell walls, which overwhelms their defenses.
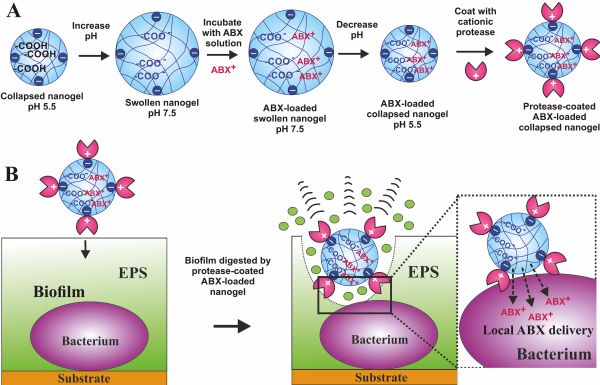
We demonstrated their effectiveness against six wound biofilm-forming bacteria, Staphylococcus aureus, Pseudomonas aeruginosa, Staphylococcus epidermidis, Klebsiella pneumoniae, Escherichia coli, and Enterococcus faecalis. We confirmed a 6-fold decrease in the biofilm mass and a substantial reduction in bacterial cell density using fluorescence, atomic force, and scanning electron microscopy. Additionally, we showed that co-treatments of ciprofloxacin and Alcalase-coated Carbopol nanogels led to a 3-log reduction in viable biofilm-forming cells when compared to ciprofloxacin treatments alone. Encapsulating an equivalent concentration of ciprofloxacin into the Alcalase-coated nanogel particles boosted their antibacterial effect much further, reducing the bacterial cell viability to below detectable amounts after 6 h of treatment.

The Alcalase-coated nanogel particles were noncytotoxic to human adult keratinocyte cells (HaCaT), inducing a very low apoptotic response in these cells. Overall, we demonstrated that the Alcalase-coated nanogels loaded with a cationic antibiotic elicit very strong biofilm-clearing effects against wound-associated biofilm-forming pathogenic bacteria. This nanotechnology approach has the potential to become a very powerful treatment of chronically infected wounds with biofilm-forming bacteria.
References
1. Weldrick, P.J., Hardman, M.J., Paunov, V.N., Enhanced Clearing of Wound-Related Pathogenic Bacterial Biofilms Using Protease-Functionalized Antibiotic Nanocarriers, ACS Appl. Mater. Interf., 11 (2019) 43902-43919.
